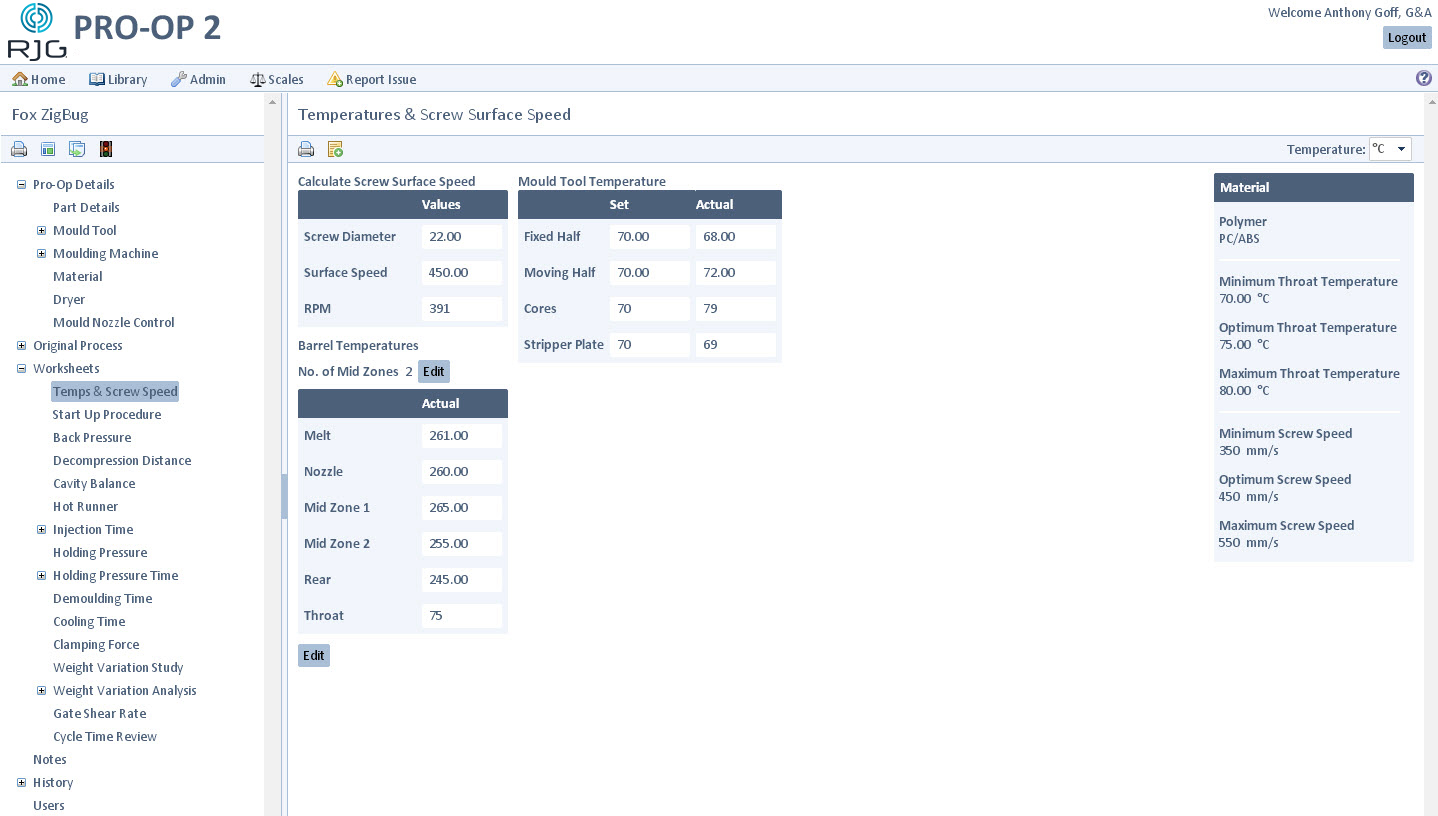
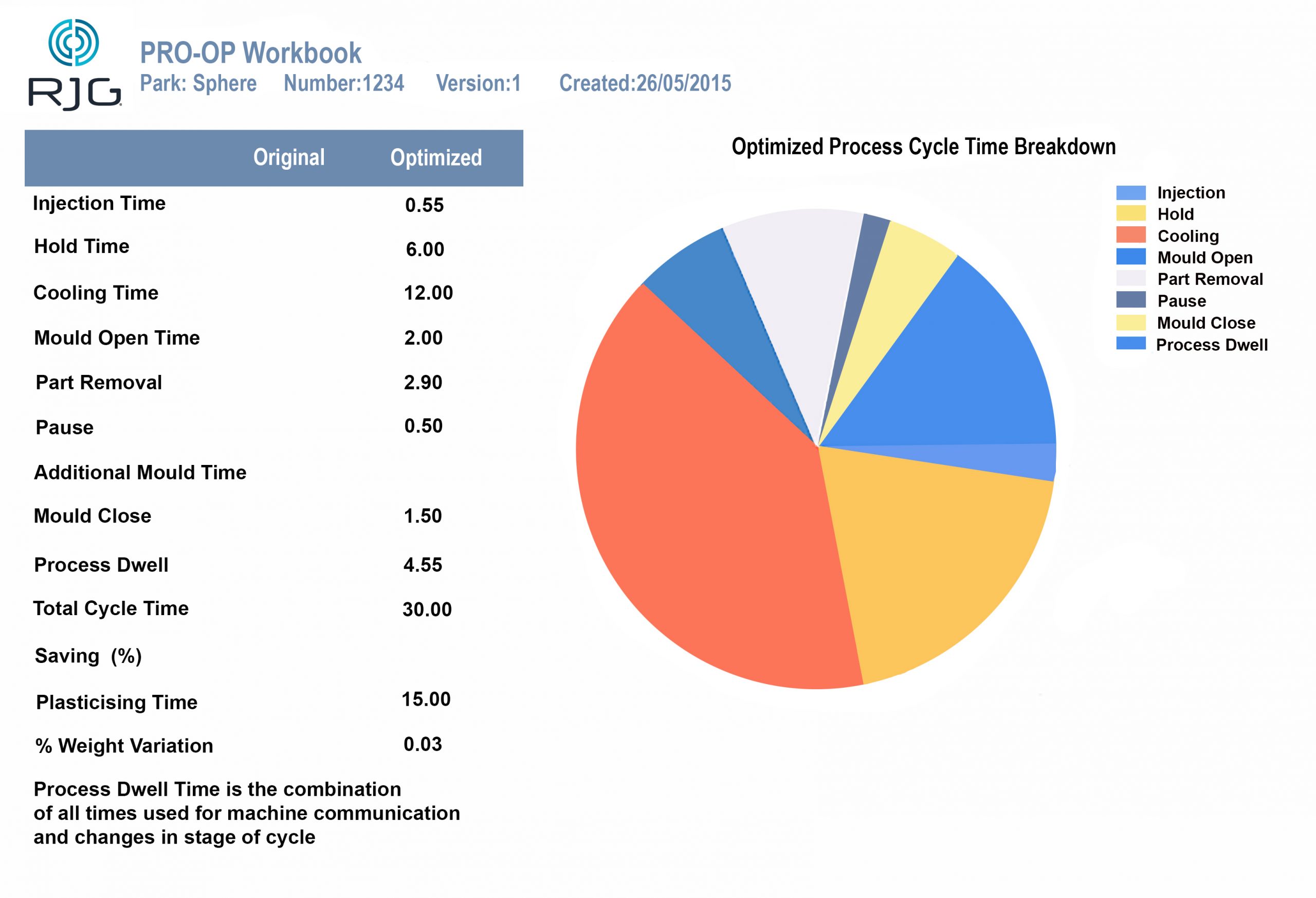
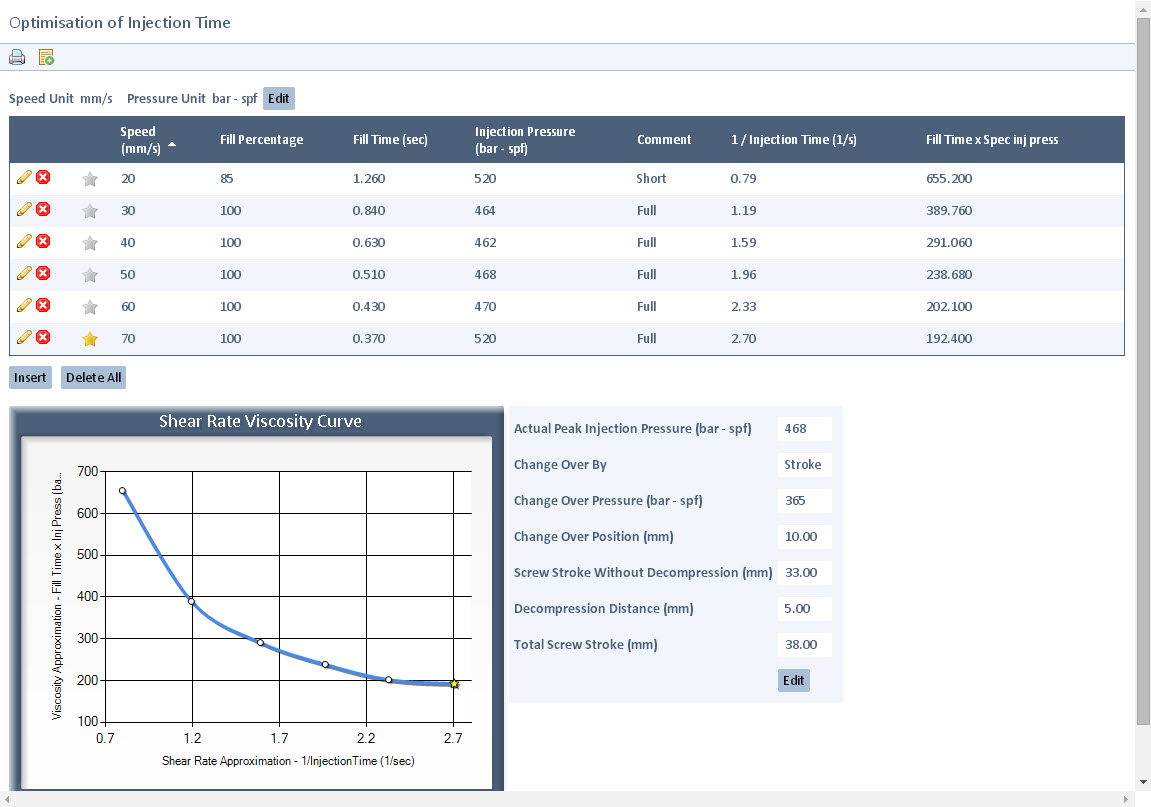
Build consistent, validated processes for optimum output
An FDA compliant software application serving all injection molding industries
Maximize accuracy and output
- Step-by-step walkthrough
- Online instruction and support
- Part repeatability
Mold with confidence
- FDA compliant validation protocol
- Complete history tracking to reduce risk
- Downloadable reports
Mold with confidence
- Self-validating system to prevent short-cutting
- Version control
- Internal approval function with email trail and notification
- Internal bug tracking and user feedback mechanism
- Ongoing technical support
- Annual functionality updates and options for bespoke customer functionality